open 10 am - 7 pm
laboratory is closed
Alexandrite

Alexandrite is a stone that belongs to the chrysoberyl family that transforms from green or blue-green to purple-red or purple when viewed under different light sources (daylight or incandescent lamp). This type of chrysoberyl was first discovered in the Urals in the 1830s, and named alexandrite in honor of the future Tsar Alexander II of Russia by Nils Adolf Erik Nordenskiöld (November 18, 1832 - August 12, 1901) in 1842.
Chemistry: BeAl2O4;
Crystal system: Rhombic, Rhombo-dipyramidal symmetry class (point symmetry group);
Color: The main color of alexandrite (green-red), like the alexandrite effect, is caused by the admixture of Cr3+, isomorphically displacing Al3+.
Hues and colors other than the classical ones are caused by an admixture of Fe3+ and/or V3+.
Identification properties
| Physical properties | |
|---|---|
| Mohs hardness: | 8-8,5 |
| Density: | 3,64 – 3,78 g/cm3 |
| Cleavage: | distinct on (011), from imperfect to poor on (100) |
| Fracture: | conchoidal to uneven |
| Optical properties | |
|---|---|
| Optical character: | anisotropic, biaxial (from negative to positive) |
| Refractive Index: | Np =1,744 – 1,748, Nm =1,747 – 1,753, Ng =1,753 – 1,758 |
| Birefringence: | 0,007 – 0,011 |
| Pleochroism: | distinct: red-yellow-green |
| Dispertion: | 0,015 (BG) |
| Luster: | vitreous, greasy, unctuous |
Inclusions and structural inhomogeneities

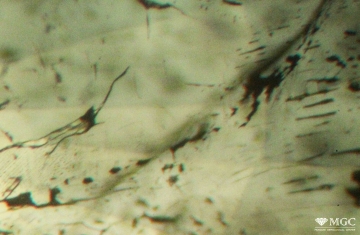

Gem basic treatments
Sometimes filling surface-reaching fractures or cavities with oil, epoxy compounds, and wax is used to improve the apparent clarity of gem materials and appearance.
Synthetic or Imitation gem materials
Synthetic alexandrite is produced both for technical purposes and for jewelry inserts.
A method of stretching from the melt, methods of zone melting, method of temperature gradient - flux (from the solution in the melt) are outlined among the methods for the alexandrite synthesis. Gas-flame method and the method of temperature gradient - hydrothermal (from aqueous solution) are used very rare.
The most commonly encountered synthetic alexandrite is as follows:
- natural "hard" minerals with alexandrite effect (garnets, corundums, spinel);
- "soft" minerals with natural or "induced" as a result of treatment with alexandrite effect (apatite, fluorite, etc.) - mainly in the form of cabochons;
- synthetic minerals with a similar color effect (fianit, garnet, spinel, corundum);
- doublets and triplets; the most frequent combinations are garnet + glass, chrysoberyl + glass, corundum + glass;
- alexandrite glass.
Inclusions and structural inhomogeneities



